Spatial-temporal analysis
To probe spatial-temporal dynamics during early development, we used SLAT to align two spatial atlases of developing mouse embryonic at E11.5 and E12.5 via Stereo-seq data (Chen. et al). We did subsample of each dataset to ~10,000 cells for the purpose of demonstration.
You need following files as input:
adata1.h5ad: E11.5 mouse embryo dataset, download from here
adata2.h5ad: E12.5 mouse embryo dataset, download from here
[1]:
import os
import scanpy as sc
import numpy as np
import pandas as pd
import torch
import scSLAT
from scSLAT.model import load_anndatas, Cal_Spatial_Net, run_SLAT, scanpy_workflow, spatial_match
from scSLAT.viz import match_3D_multi, hist, Sankey, match_3D_celltype, Sankey
from scSLAT.metrics import region_statistics
[2]:
sc.set_figure_params(dpi=100, dpi_save=150)
[3]:
adata1 = sc.read_h5ad('E115_Stereo.h5ad')
adata2 = sc.read_h5ad('E125_Stereo.h5ad')
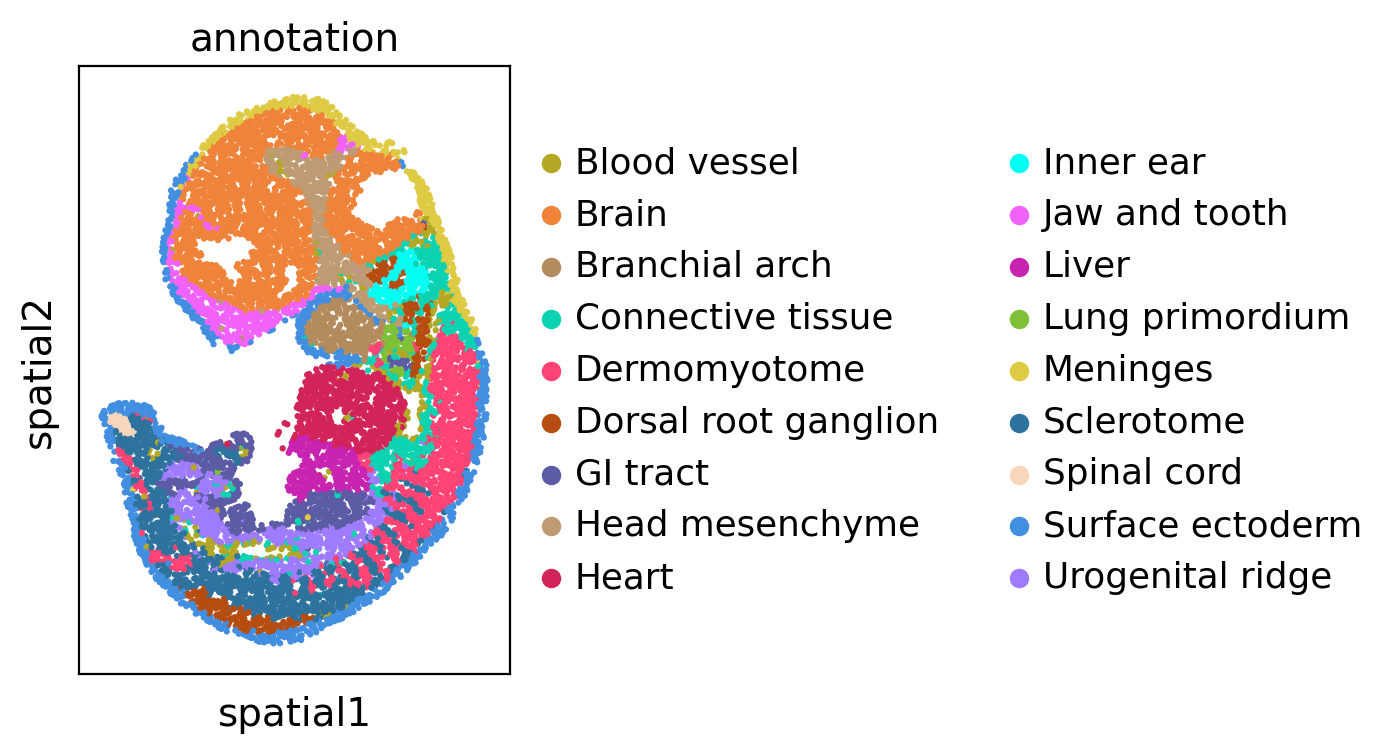
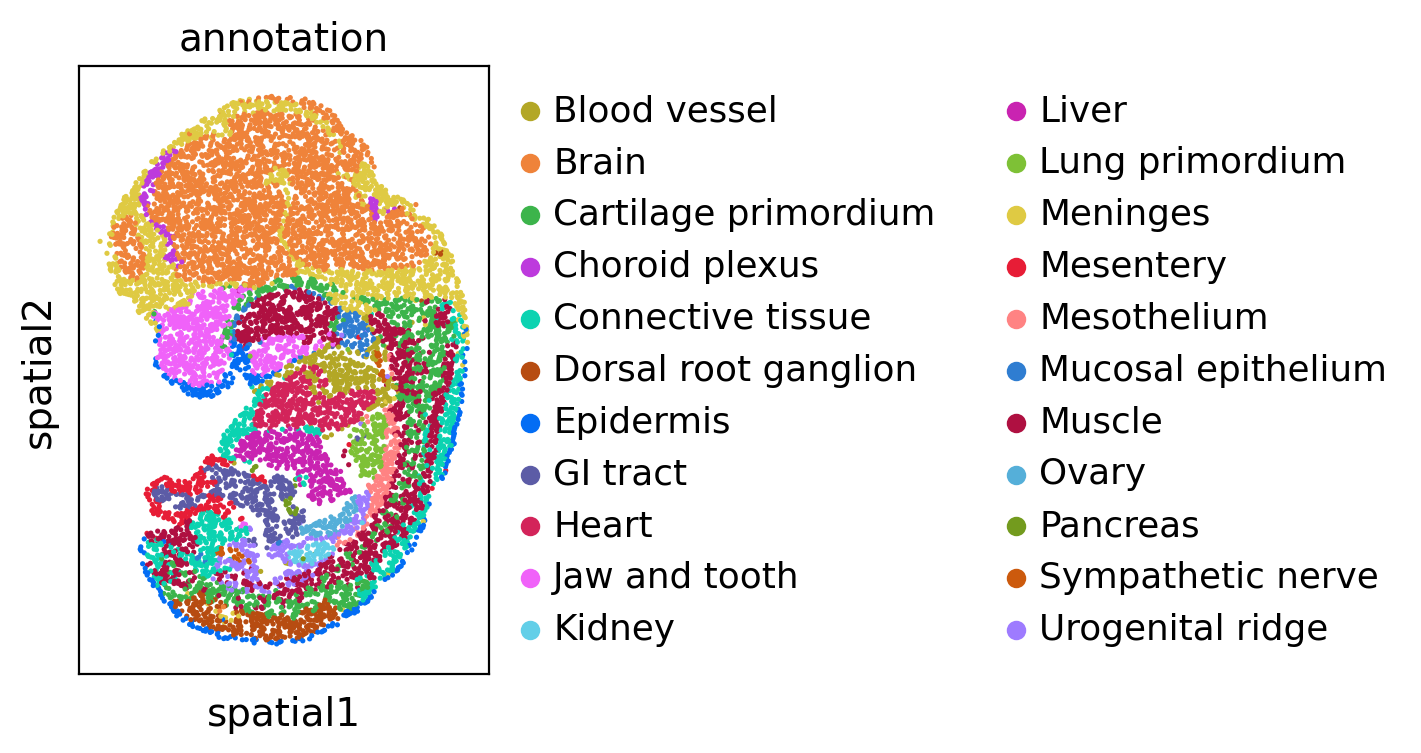
We visualize the dataset and colored by cell type annotation.
[4]:
sc.pl.spatial(adata1, color='annotation', spot_size=3)
sc.pl.spatial(adata2, color='annotation', spot_size=3)
Run SLAT
Then we run SLAT as usual
[5]:
Cal_Spatial_Net(adata1, k_cutoff=20, model='KNN')
Cal_Spatial_Net(adata2, k_cutoff=20, model='KNN')
edges, features = load_anndatas([adata1, adata2], feature='DPCA', check_order=False)
Calculating spatial neighbor graph ...
The graph contains 537308 edges, 10000 cells.
53.7308 neighbors per cell on average.
Calculating spatial neighbor graph ...
The graph contains 539939 edges, 10001 cells.
53.98850114988501 neighbors per cell on average.
Use DPCA feature to format graph
/rd2/user/xiacr/SLAT/conda/lib/python3.8/site-packages/anndata/_core/anndata.py:1785: FutureWarning: X.dtype being converted to np.float32 from float64. In the next version of anndata (0.9) conversion will not be automatic. Pass dtype explicitly to avoid this warning. Pass `AnnData(X, dtype=X.dtype, ...)` to get the future behavour.
[AnnData(sparse.csr_matrix(a.shape), obs=a.obs) for a in all_adatas],
/rd2/user/xiacr/SLAT/conda/lib/python3.8/site-packages/scanpy/preprocessing/_normalization.py:170: UserWarning: Received a view of an AnnData. Making a copy.
view_to_actual(adata)
/rd2/user/xiacr/SLAT/conda/lib/python3.8/site-packages/scanpy/preprocessing/_normalization.py:170: FutureWarning: X.dtype being converted to np.float32 from float64. In the next version of anndata (0.9) conversion will not be automatic. Pass dtype explicitly to avoid this warning. Pass `AnnData(X, dtype=X.dtype, ...)` to get the future behavour.
view_to_actual(adata)
/rd2/user/xiacr/SLAT/conda/lib/python3.8/site-packages/scanpy/preprocessing/_simple.py:843: UserWarning: Received a view of an AnnData. Making a copy.
view_to_actual(adata)
/rd2/user/xiacr/SLAT/conda/lib/python3.8/site-packages/scanpy/preprocessing/_simple.py:843: UserWarning: Received a view of an AnnData. Making a copy.
view_to_actual(adata)
Warning! Dual PCA is using GPU, which may lead to OUT OF GPU MEMORY in big dataset!
[6]:
embd0, embd1, time = run_SLAT(features, edges, LGCN_layer=5)
Choose GPU:5 as device
Running
---------- epochs: 1 ----------
---------- epochs: 2 ----------
---------- epochs: 3 ----------
---------- epochs: 4 ----------
---------- epochs: 5 ----------
---------- epochs: 6 ----------
Training model time: 1.83
[7]:
best, index, distance = spatial_match([embd0, embd1], reorder=False, adatas=[adata1,adata2])
[8]:
adata1_df = pd.DataFrame({'index':range(embd0.shape[0]),
'x': adata1.obsm['spatial'][:,0],
'y': adata1.obsm['spatial'][:,1],
'celltype':adata1.obs['annotation']})
adata2_df = pd.DataFrame({'index':range(embd1.shape[0]),
'x': adata2.obsm['spatial'][:,0],
'y': adata2.obsm['spatial'][:,1],
'celltype':adata2.obs['annotation']})
matching = np.array([range(index.shape[0]), best])
best_match = distance[:,0]
region_statistics(best_match, start=0.5, number_of_interval=10)
0.500~0.550 11 0.110%
0.550~0.600 46 0.460%
0.600~0.650 234 2.340%
0.650~0.700 640 6.399%
0.700~0.750 1187 11.869%
0.750~0.800 1553 15.528%
0.800~0.850 1830 18.298%
0.850~0.900 1821 18.208%
0.900~0.950 2039 20.388%
0.950~1.000 637 6.369%
Visualization of alignment
[9]:
multi_align = match_3D_multi(adata1_df, adata2_df, matching, meta='celltype',
scale_coordinate=True, subsample_size=300, exchange_xy=False)
multi_align.draw_3D(size=[7, 8], line_width =1, point_size=[0.8,0.8], hide_axis=True, show_error=False)
dataset1: 18 cell types; dataset2: 22 cell types;
Total :29 celltypes; Overlap: 11 cell types
Not overlap :[['Spinal cord', 'Branchial arch', 'Sclerotome', 'Inner ear', 'Dermomyotome', 'Surface ectoderm', 'Head mesenchyme', 'Sympathetic nerve', 'Ovary', 'Muscle', 'Kidney', 'Mucosal epithelium', 'Mesentery', 'Mesothelium', 'Choroid plexus', 'Cartilage primordium', 'Epidermis', 'Pancreas']]
Subsample 300 cell pairs from 10001
/rd2/user/xiacr/SLAT/scSLAT/viz/multi_dataset.py:204: FutureWarning: The series.append method is deprecated and will be removed from pandas in a future version. Use pandas.concat instead.
self.celltypes = set(self.dataset_A[meta].append(self.dataset_B[meta]))

[10]:
adata2.obs['low_quality_index']= best_match
adata2.obs['low_quality_index'] = adata2.obs['low_quality_index'].astype(float)
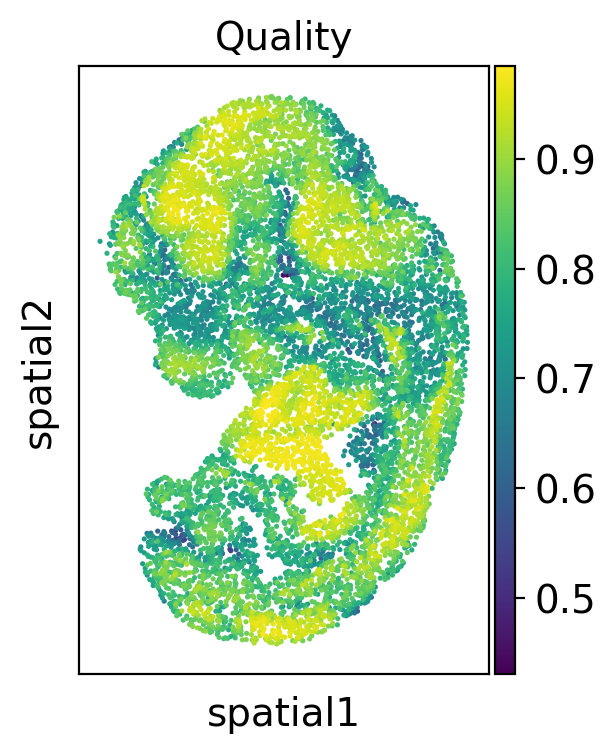
Then we check the alignment quality of the whole slide
[11]:
sc.pl.spatial(adata2, color='low_quality_index', spot_size=3, title='Quality')
We use a Sankey diagram to show the correspondence between cell types at different stages of development
[12]:
adata2_df['target_celltype'] = adata1_df.iloc[matching[1,:],:]['celltype'].to_list()
[13]:
matching_table = adata2_df.groupby(['celltype', 'target_celltype']).size().unstack(fill_value=0)
[14]:
Sankey(matching_table, layout=[1000,800], font_size=15)
Focus on developing Kidney
We highlighted the “Kidney” cells in E12.5 and their aligned precursor cells in E11.5 in alignment results. Consistent with our biological priors, the precursors of the kidney are the mesonephros and the metanephros
[15]:
kidney_align = match_3D_celltype(adata1_df, adata2_df, matching, meta='celltype', highlight_celltype = [['Urogenital ridge'],['Kidney']],
subsample_size=10000, highlight_line = ['blue'], scale_coordinate = True )
kidney_align.draw_3D(size= [10, 12], line_width =0.8, point_size=[0.6,0.6], hide_axis=True)
/rd2/user/xiacr/SLAT/scSLAT/viz/multi_dataset.py:204: FutureWarning:
The series.append method is deprecated and will be removed from pandas in a future version. Use pandas.concat instead.
dataset1: 18 cell types; dataset2: 22 cell types;
Total :29 celltypes; Overlap: 11 cell types
Not overlap :[['Spinal cord', 'Branchial arch', 'Sclerotome', 'Inner ear', 'Dermomyotome', 'Surface ectoderm', 'Head mesenchyme', 'Sympathetic nerve', 'Ovary', 'Muscle', 'Kidney', 'Mucosal epithelium', 'Mesentery', 'Mesothelium', 'Choroid plexus', 'Cartilage primordium', 'Epidermis', 'Pancreas']]
Subsample 10000 cell pairs from 10001

Focus on developing Ovary
Then we focus on another organ: ‘Ovary’, and found ovary only has single spatial origin. It is interesting that precursors of ovary are spatially close to the mesonephros (see Kidney part), because mammalian ovary originates from the regressed mesonephros.
[16]:
Ovary_align = match_3D_celltype(adata1_df, adata2_df, matching, meta='celltype', highlight_celltype = [['Urogenital ridge'],['Ovary']],
subsample_size=10000, highlight_line = ['blue'], scale_coordinate = True)
Ovary_align.draw_3D(size= [10, 12], line_width =0.8, point_size=[1.2,1.2], hide_axis=True)
/rd2/user/xiacr/SLAT/scSLAT/viz/multi_dataset.py:204: FutureWarning:
The series.append method is deprecated and will be removed from pandas in a future version. Use pandas.concat instead.
dataset1: 18 cell types; dataset2: 22 cell types;
Total :29 celltypes; Overlap: 11 cell types
Not overlap :[['Spinal cord', 'Branchial arch', 'Sclerotome', 'Inner ear', 'Dermomyotome', 'Surface ectoderm', 'Head mesenchyme', 'Sympathetic nerve', 'Ovary', 'Muscle', 'Kidney', 'Mucosal epithelium', 'Mesentery', 'Mesothelium', 'Choroid plexus', 'Cartilage primordium', 'Epidermis', 'Pancreas']]
Subsample 10000 cell pairs from 10001
